Peptide storage
Lyophilized (recommended): For maximum stability, peptides should be stored lyophilized at -20°C. When using refrigerated peptide, allow the vial to warm to room temperature for 30 min in a dessicator before opening the tube. Many peptides are hygroscopic and may take up some atmospheric water upon opening the vial.
Dissolved peptides: Peptides in solution are much less stable then when lyophilized. And we recommend the following for optimized stability and storage of dissolved peptides:
• Store at -20°C in a sterile slightly acidic, buffered solution (pH 5-7).
• Aliquot your stock solution and avoid repeated thawing and freezing cycles.
Peptide dissolution
The solubility of peptides in aqueous solutions greatly depends on primary and secondary structures. In addition many modifications may affect solubility. For exemple C-amide and N-acetyl termini will significantly decrease the solubility of any sequence. For purification purposes, most peptides would dissolve in polar solvents (acetonitrile, methanol, propanol, DMF, DMSO), and some will affect retention times in reversed-phase LC.
The following experimental approaches will help you reconstitute your peptides.
Always use sterile water to dissolve peptides, as most bacteria will hydrolyze peptides very efficiently. Generally speaking it is recommended to first dissolve your peptide in water and further dilute with buffered /salts working solution. The recommended concentration for peptides with unknown solubility is 1-10 mg/ml.
For difficult to dissolve peptides, the following basic rules may help, with or without a brief sonication.
• Basic peptide (presence of lys, arg…): one may try 1-10% acetic acid.
• Acidic peptide (presence of glu, asp,…): one may try 1% ammonium hydroxide or 1-10% ammonium bicarbonate.
• In case of aggregation and/or hydrophobic peptides (aliphatic chains or aromatic amino acids) one may introduce organic polar solvents to aid breaking hydrophobic intermolecular interactions. While several solvents would work, we recommend DMSO/water or DMF/water. Once fully solvated, you may dilute your peptide using sterile water or buffered aqueous solutions to reach the working concentration.
Cysteine, methionine, tryptophan are prone to oxidation and should be dissolved and stored in oxygen-free water, obtained by bubbling nitrogen, helium or argon in water before dissolution.
KLH conjugates may prove difficult to dissolve in and it may take a long time to fully hydrate and still exhibit a turbid solution.
Sequence design
Indicated below are a few empirical rules designed to increase our chances to prepare your products under optimal conditions. These rules are derived from the basic properties of the monomers.
• Peptide length
• Hydrophobic residues
Less than 50% of total aa and less than 4-5 in a row. Beta-strands formation between peptide chains during elongation may occur when long stretches of hydrophobic residues are present. The latter will often result in poor yields.
• Reactive amino-acids
Avoid multiple cys, met and trp residues.
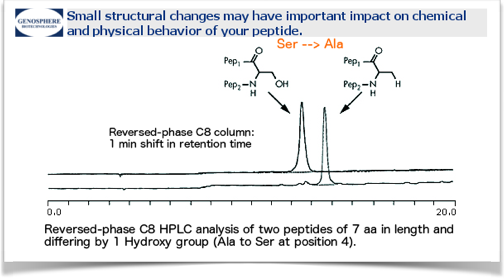
One illustration is shown in the next figure where a slight local change in peptide structure e.g., 1 internal aa, can have significant incidence on hydrophobicity of the molecule as a whole.